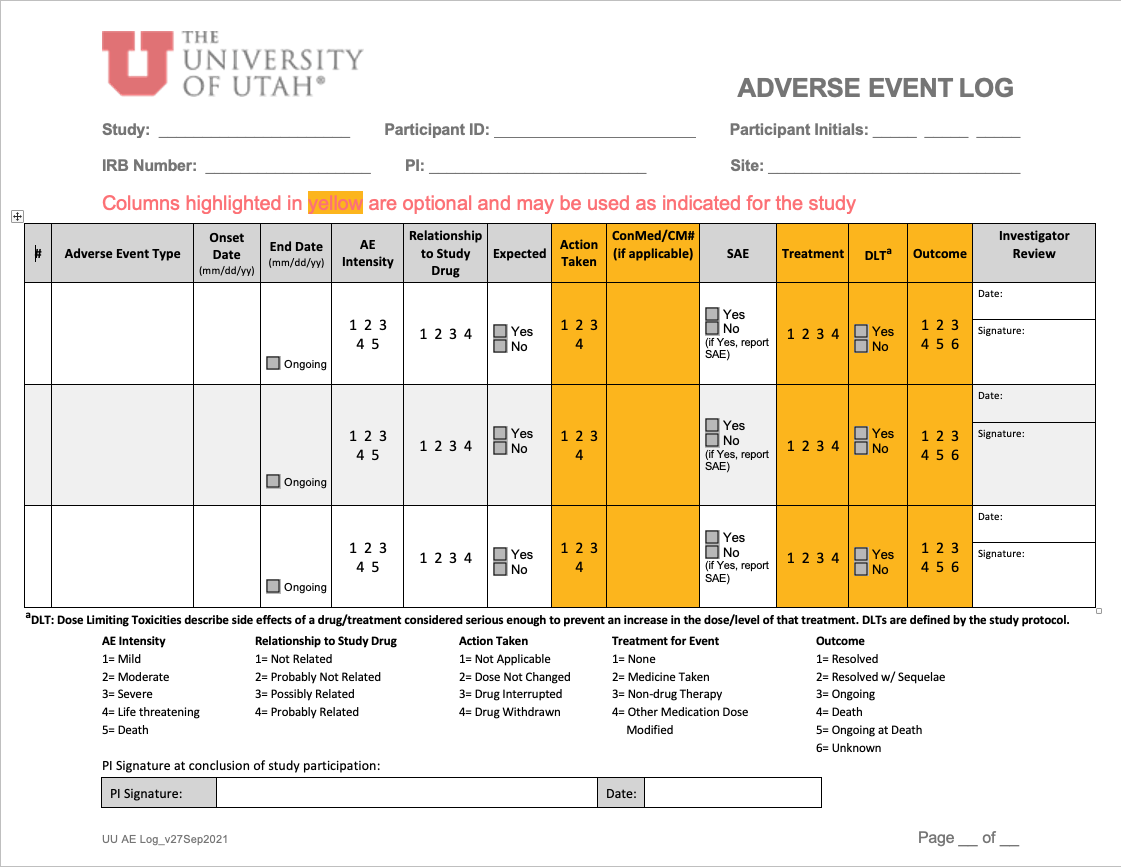
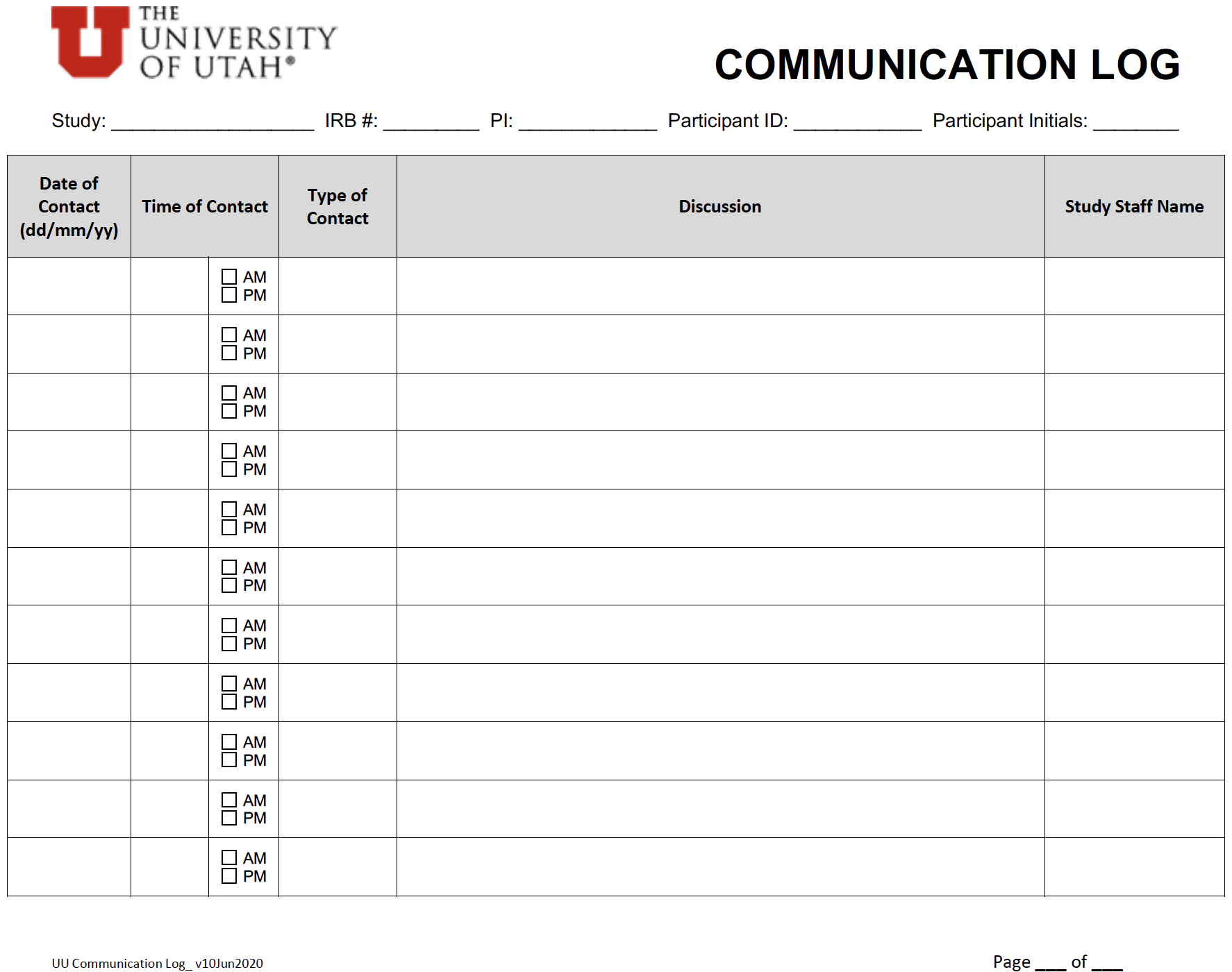
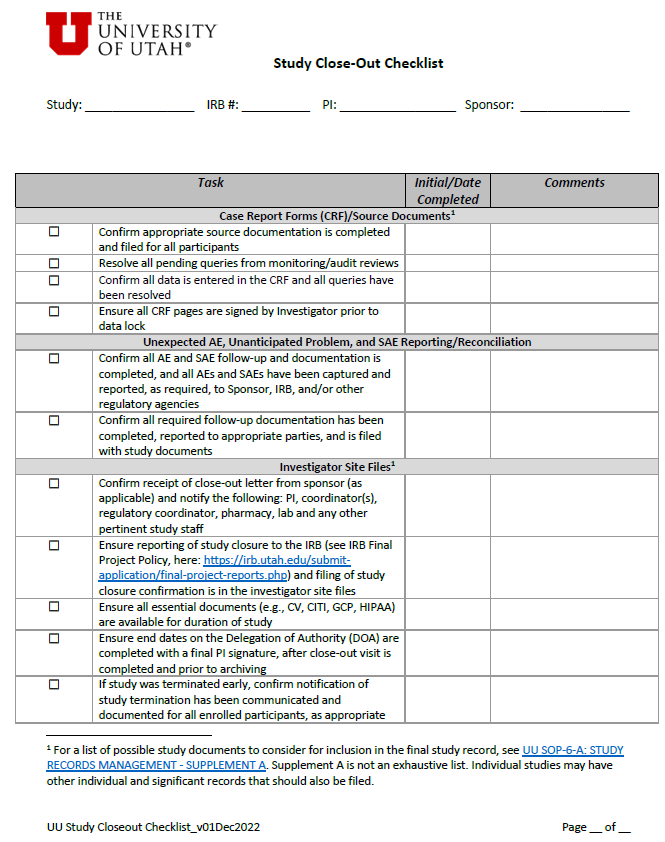
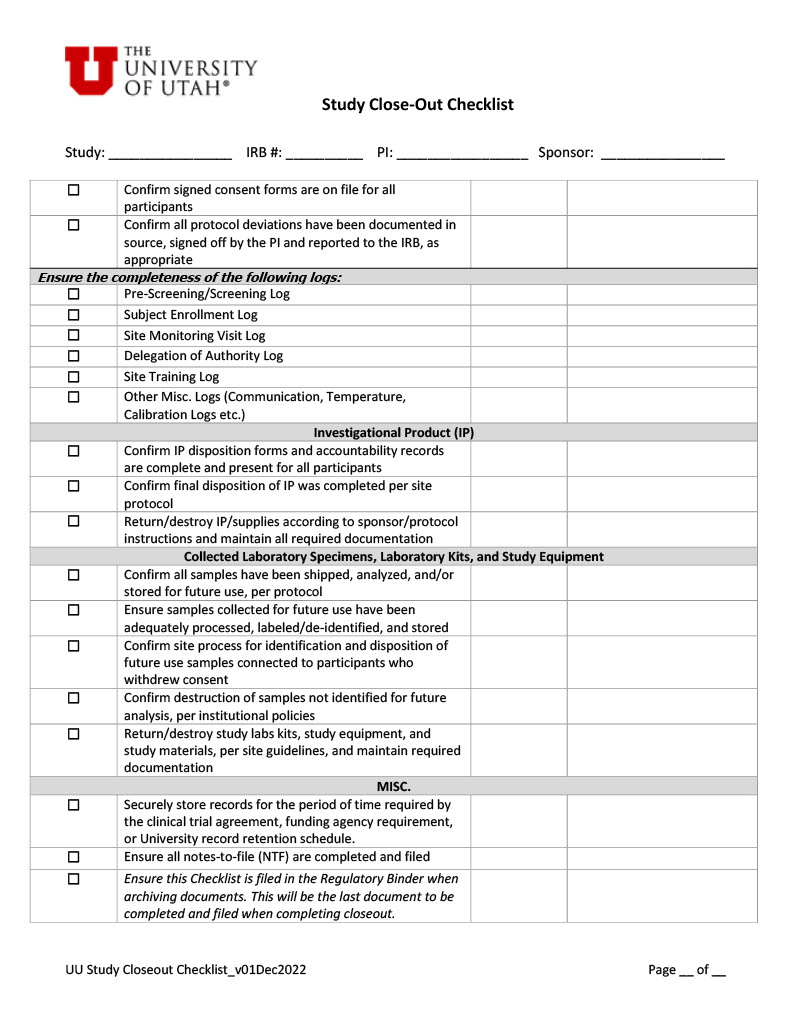
OQC Toolkit
Templates to assist research teams with conducting, tracking and/or monitoring research activities.
These documents are available for research teams to download and adapt for their own use.
Template Title |
Version Date |
|---|---|
|
September 2021 |
|
|
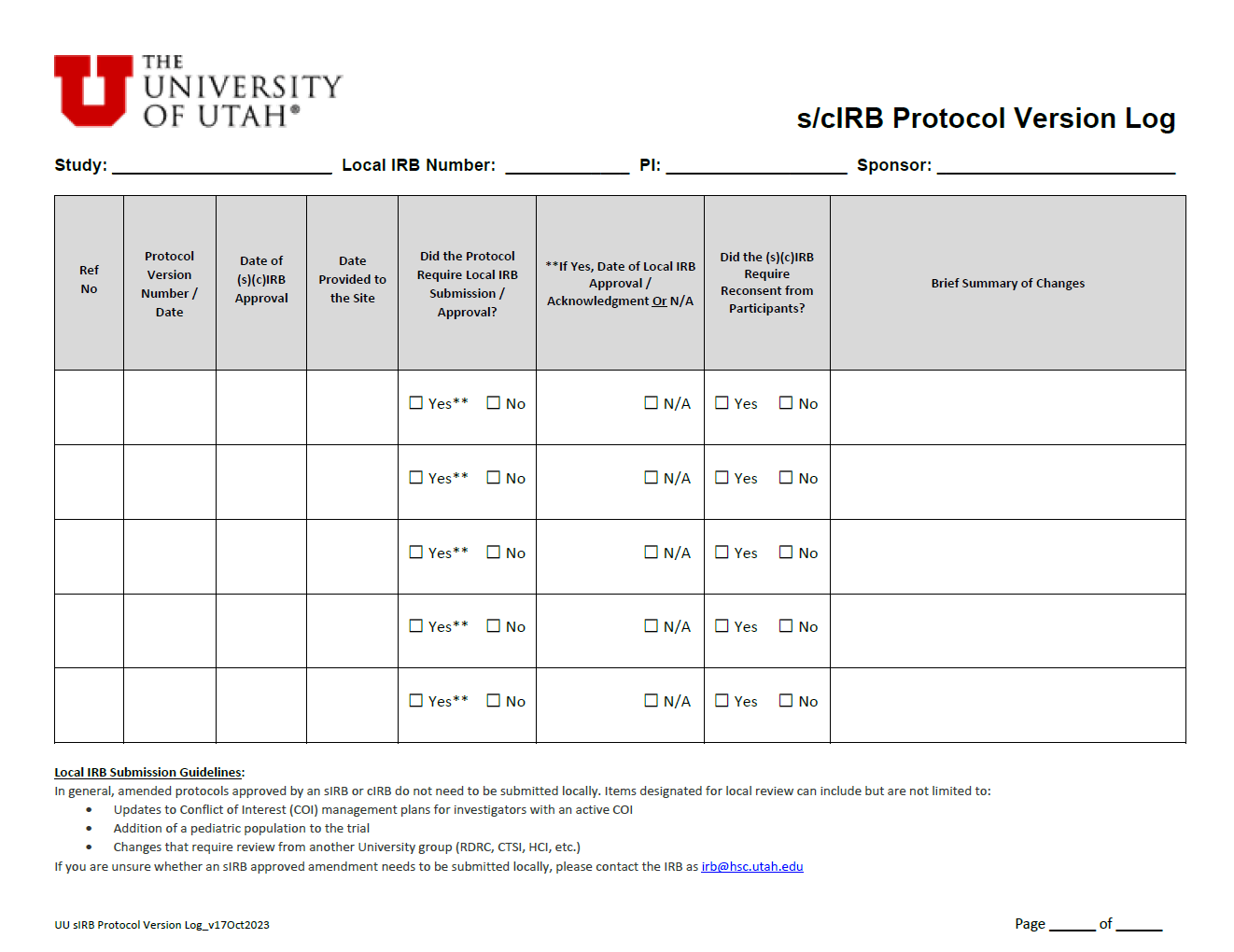
Central IRB Protocol & Consent Version Logs |
October 2023 |
|
June 2020 |
|
|
December 2022 |
|
|
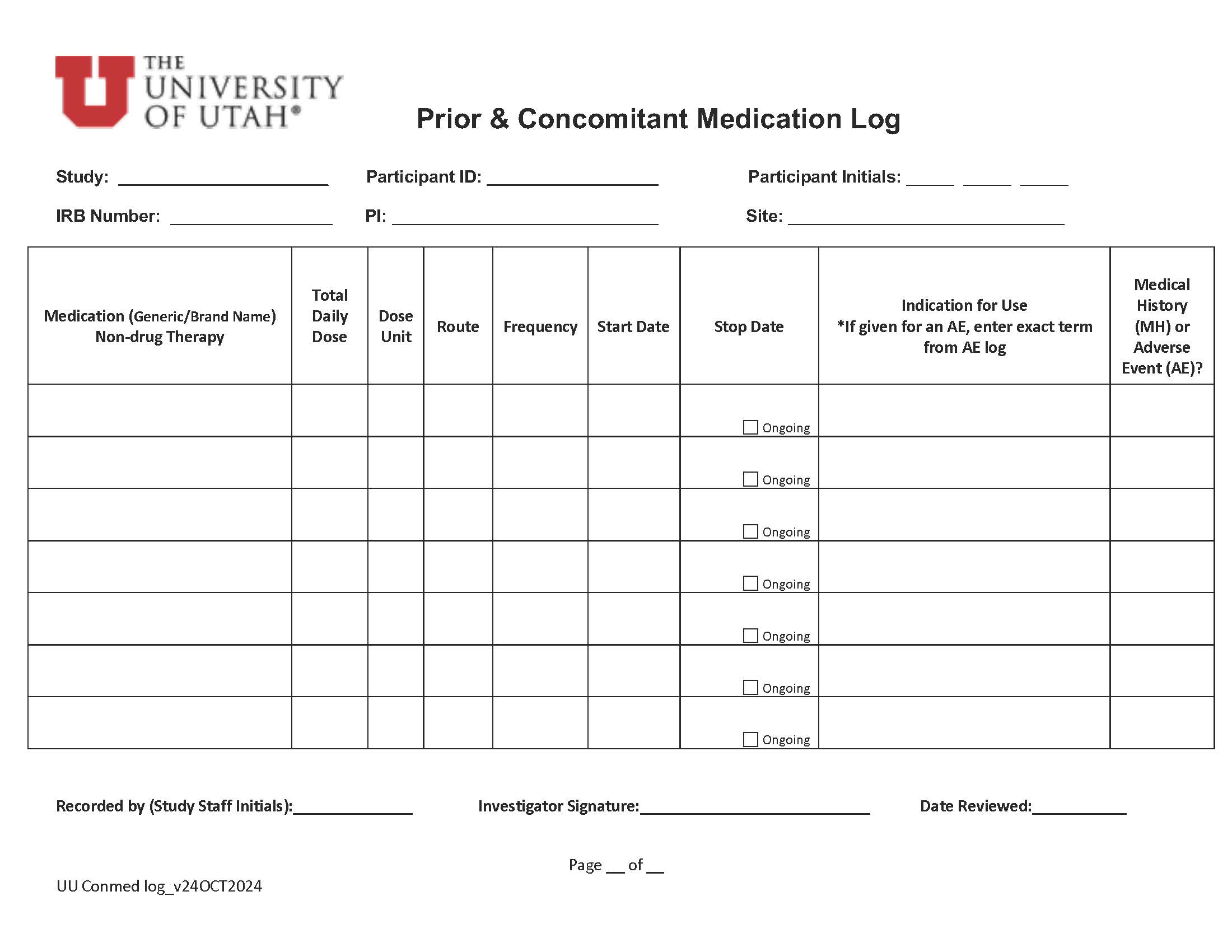
October 2024 |
|
|
June 2020 |
|
|
June 2020 |
|
|
June 2020 |
|
|
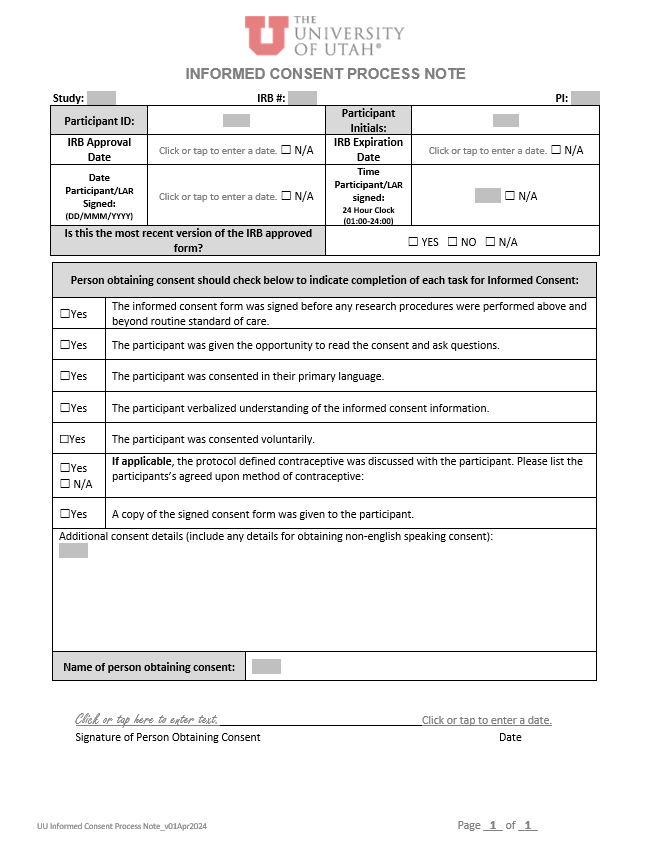
April 2024 |
|
|
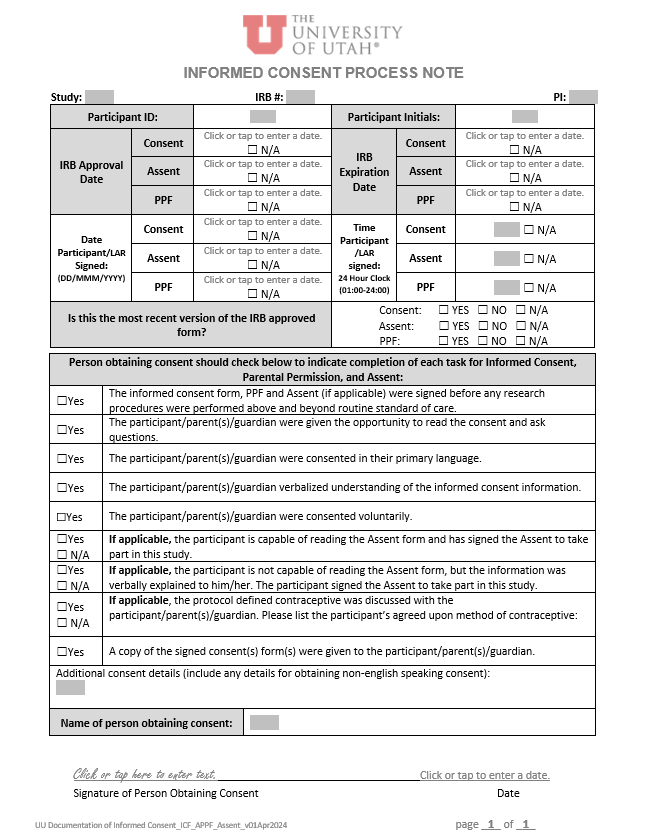
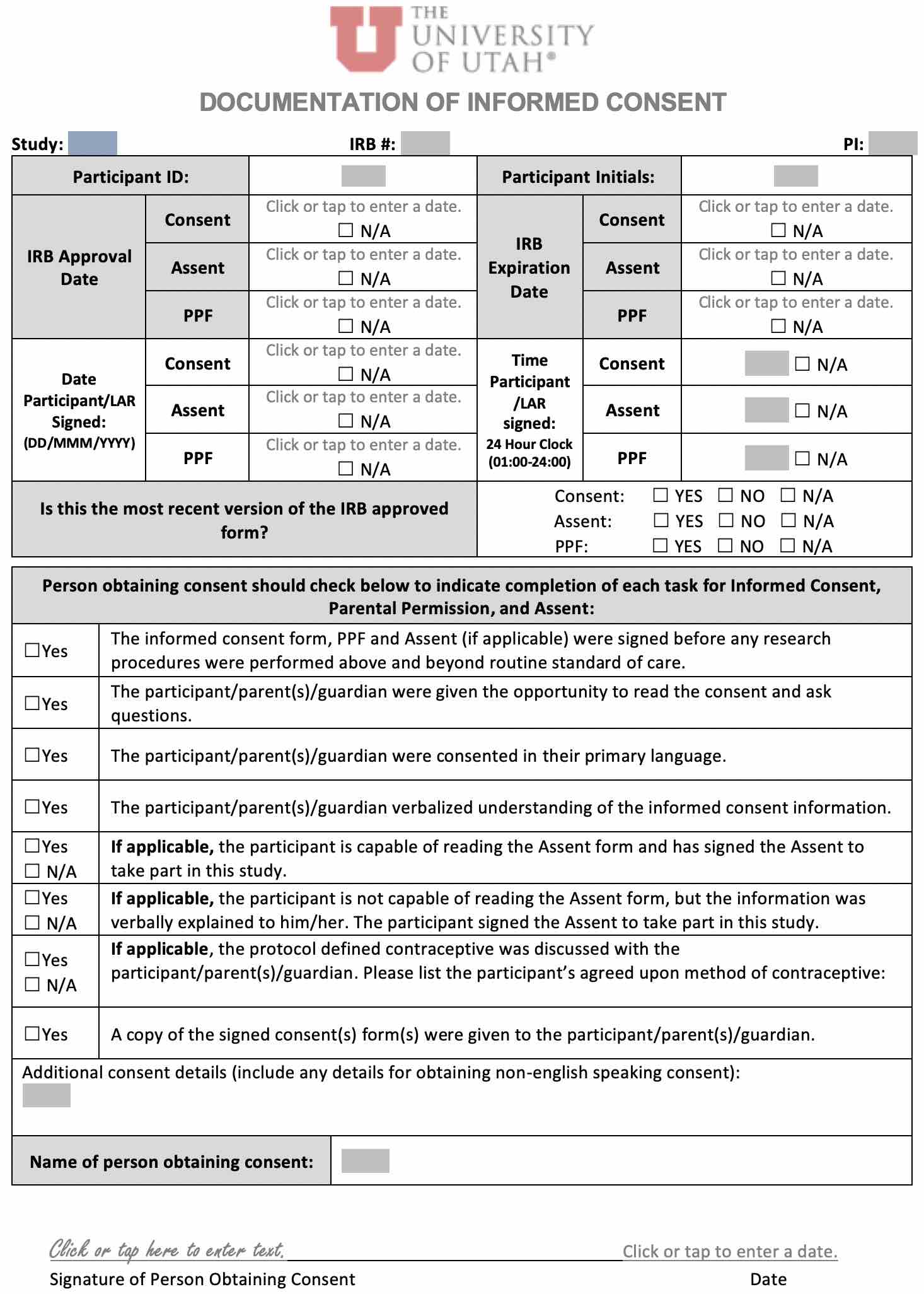
Informed Consent Process Note - Consent, Assent, Parental Permission |
April 2024 |
|
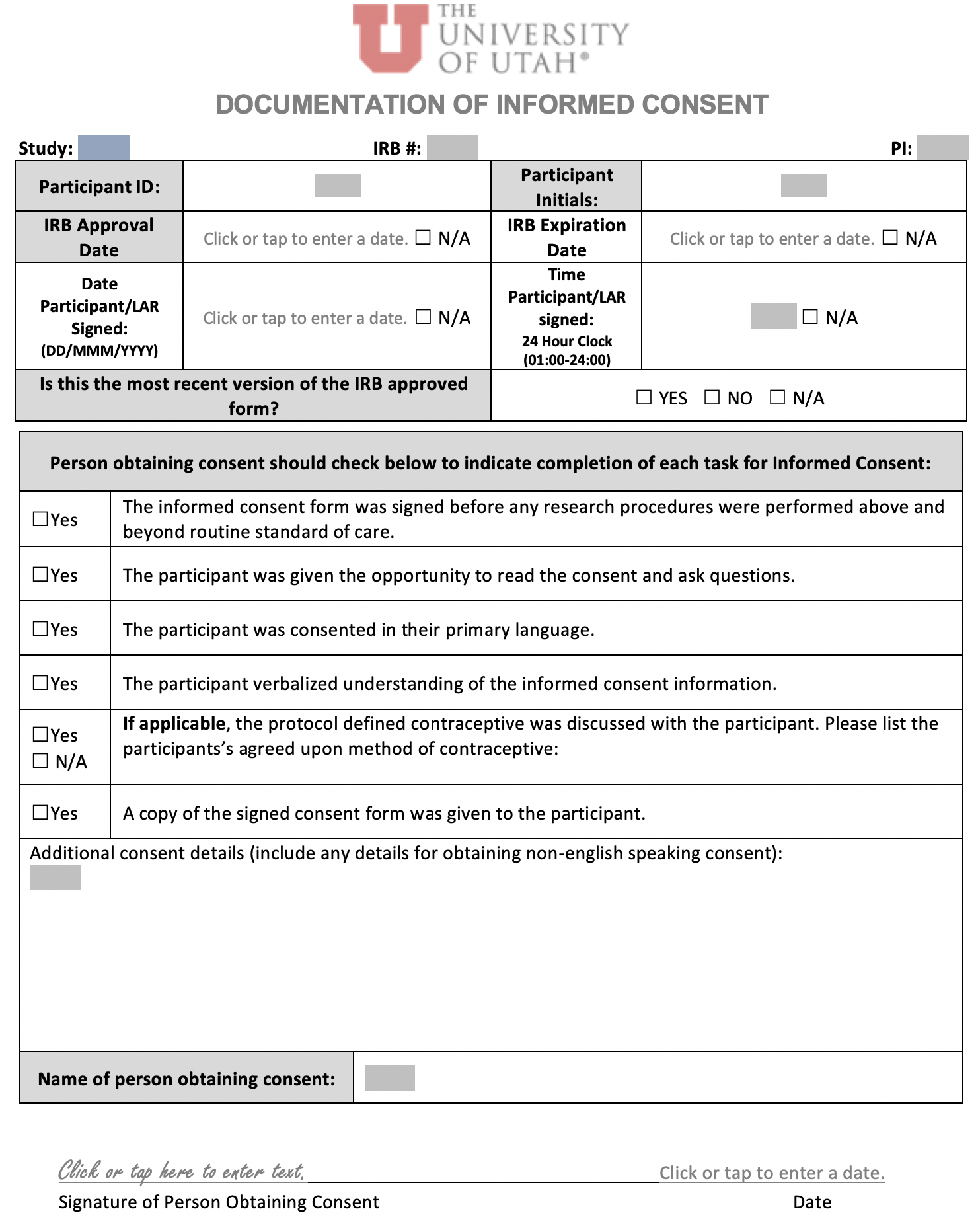
Informed Consent Process Note - Non-English Speaking Participants |
June 2024 |
|
Material Transfer Agreements (MTA) and/or Data Transfer Agreements (DTA) Checklist |
September 2021 |
|
June 2020 |
|
|
June 2020 |
|
|
June 2020 |
|
|
June 2020 |
|
|
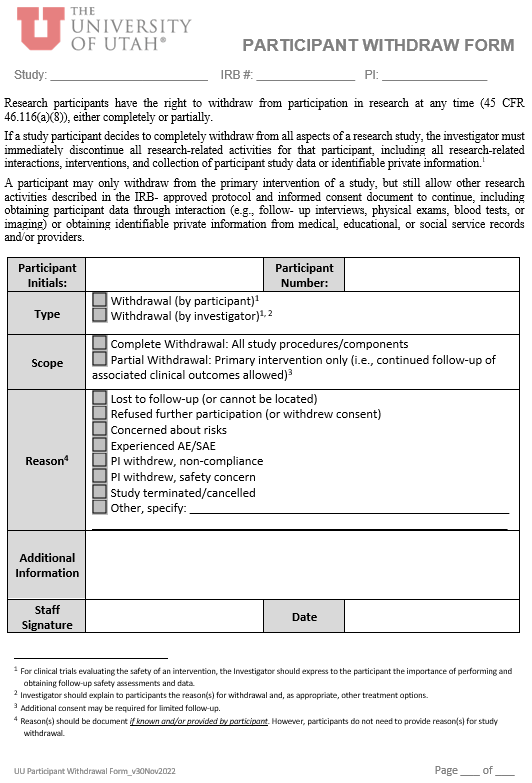
Participant Withdrawal Form Template |
November 2022 |
|
June 2020 |
|
|
June 2020 |
|
|
April 2022 |
|
|
February 2026 |
|
|
June 2020 |
|
|
June 2020 |
|
|
November 2021 |
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Preview
Yes. Investigators and researchers are free to add/remove fields on these templates in order to meet the needs of their specific studies. Researchers should use these templates as guidance for the type of data that should be collected and documented, and feel empowered to adapt the contents where needed. For example:
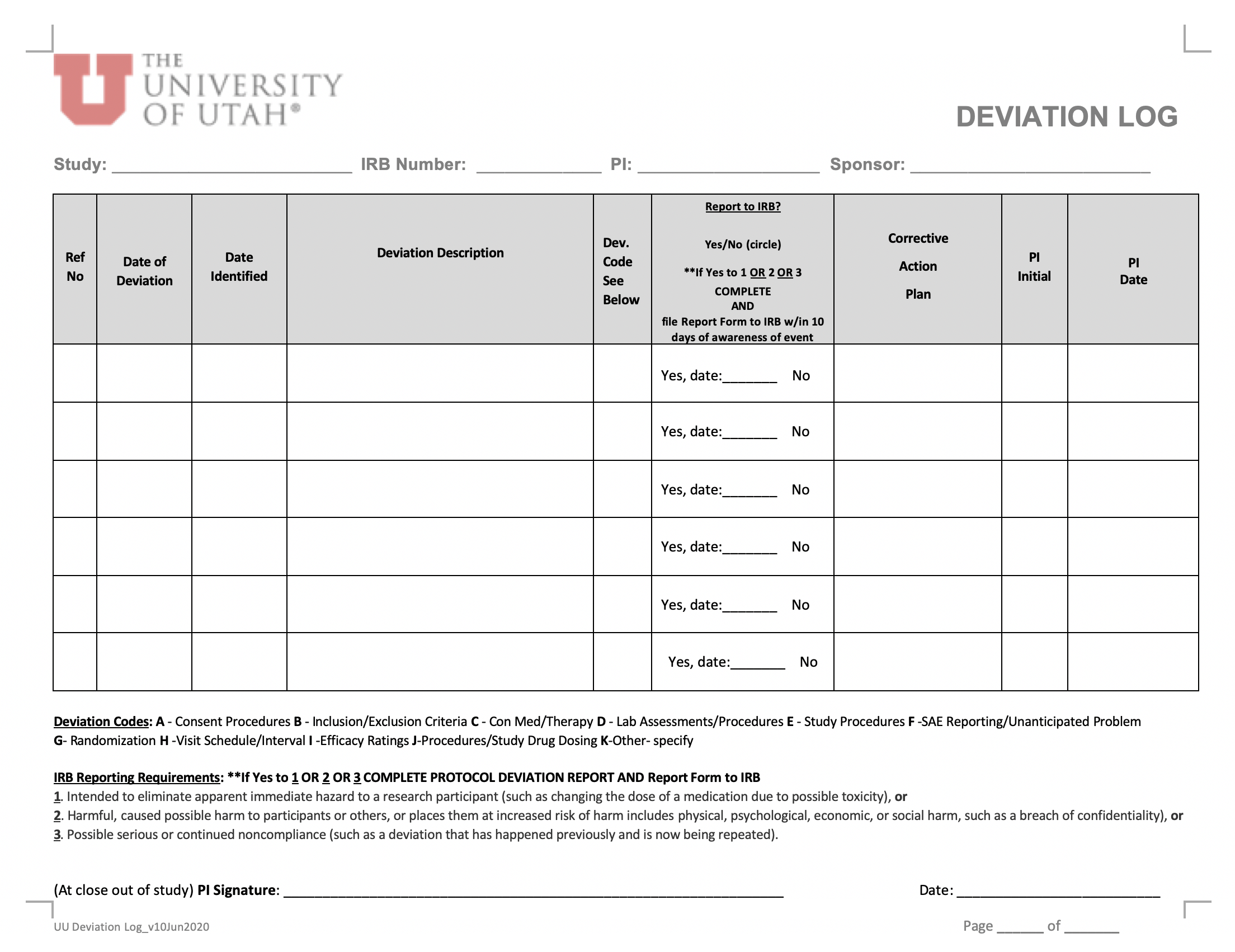
- for studies with an external sponsor, a column might be added to the “Deviation Log” to document if the deviation was reported to the study sponsor.
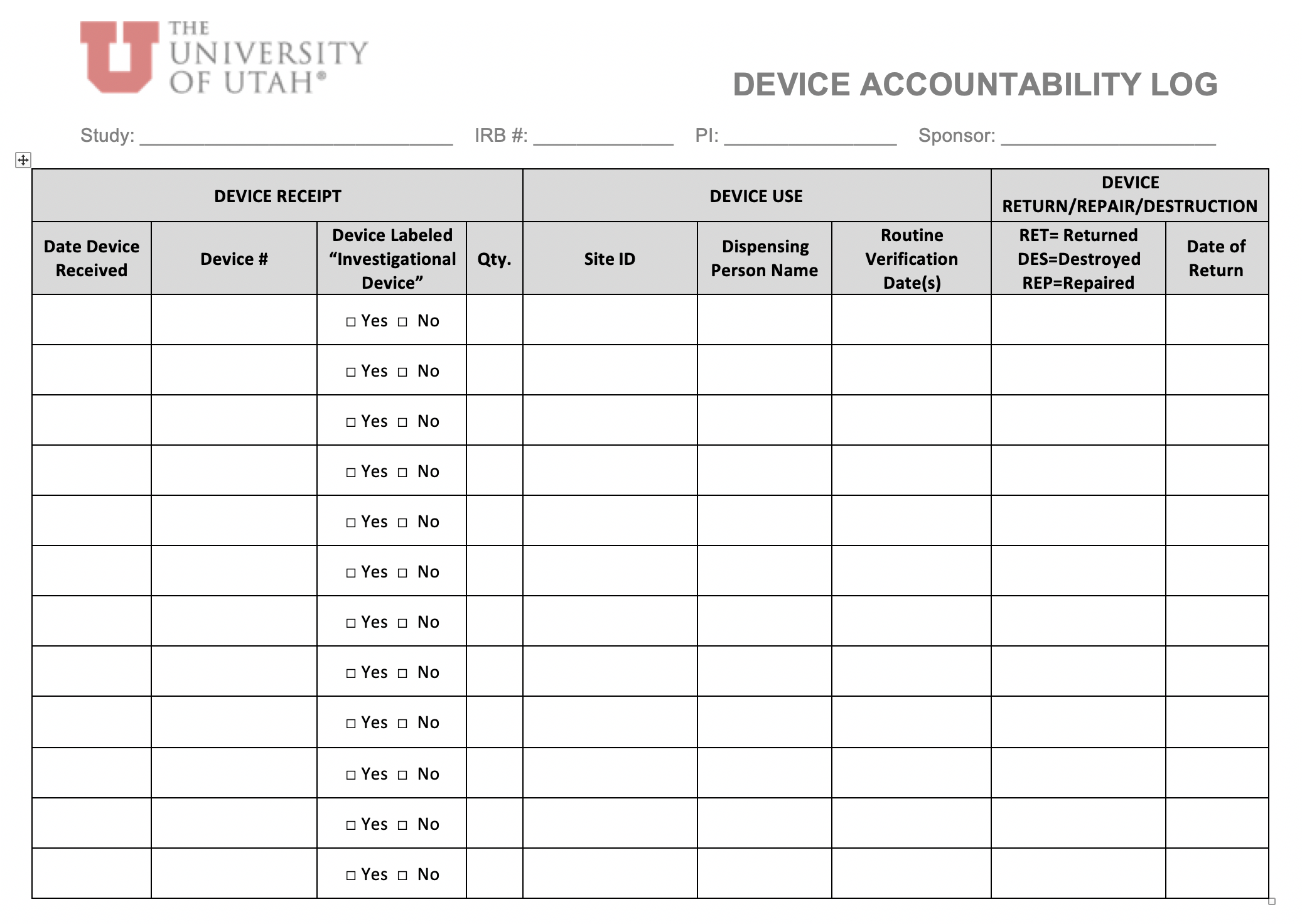
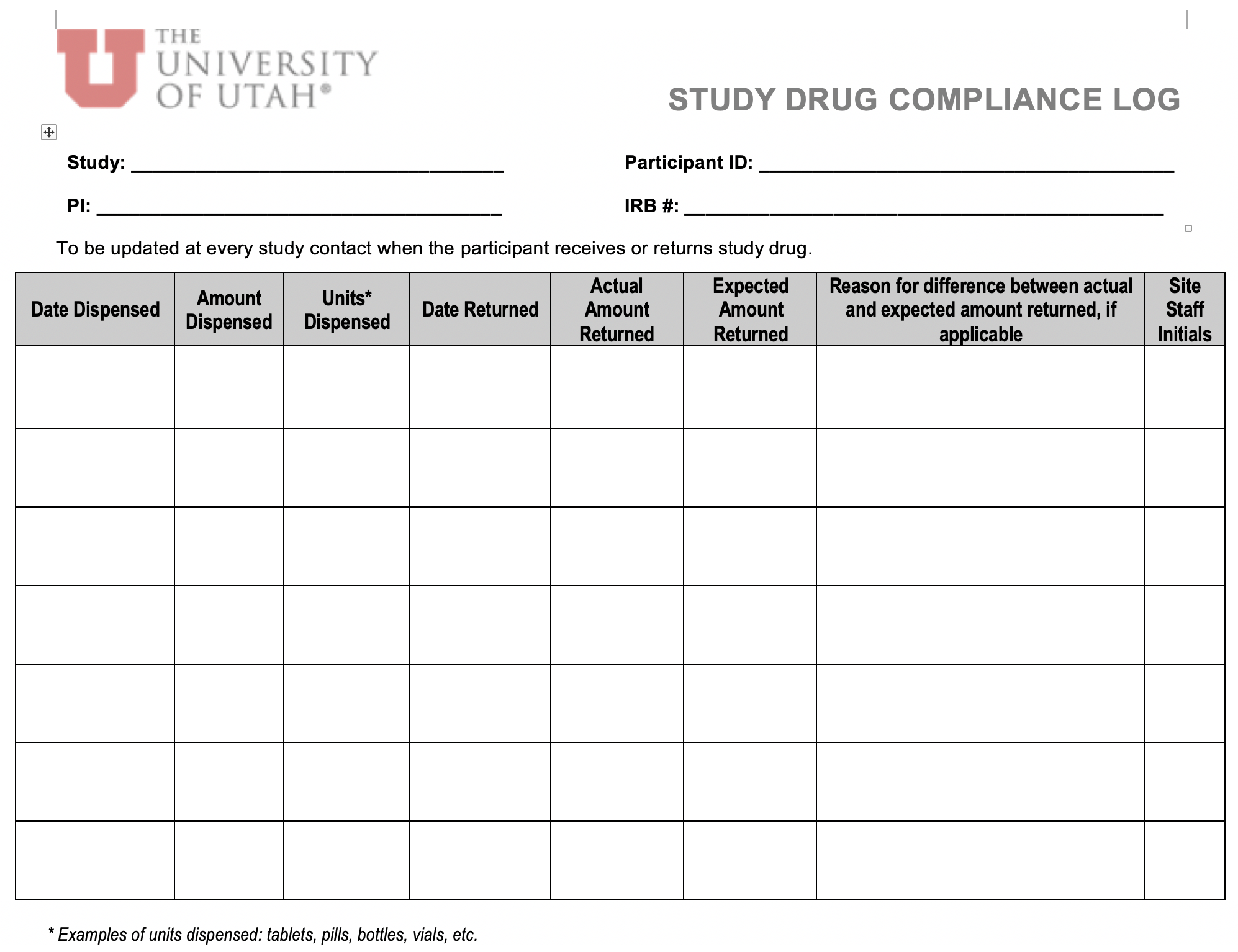
- for protocols that do not include a study drug, tasks relating to drug accountability and dispensing (i.e., #14-15) could be removed from the “Study Delegation Log”.
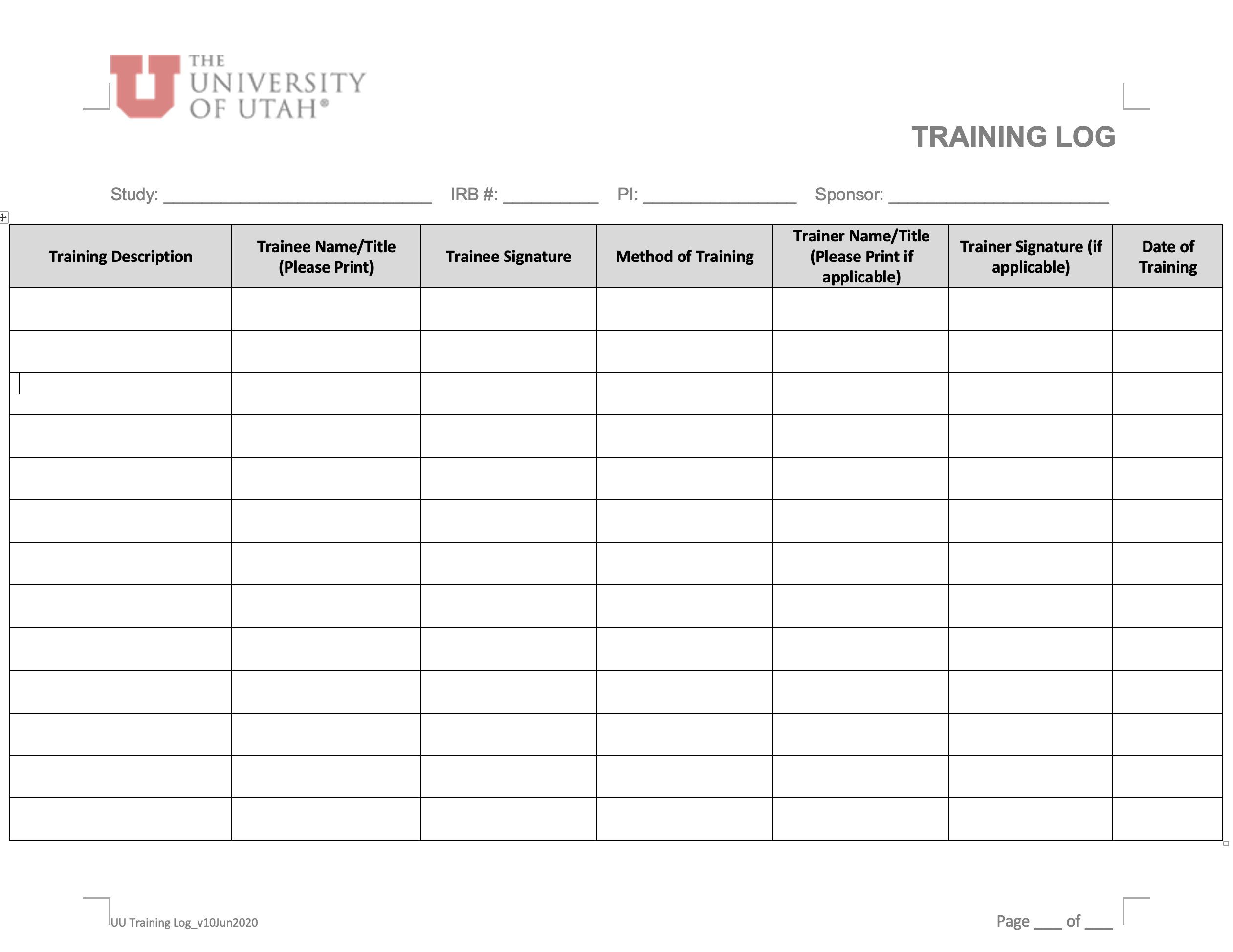
- For all study types, a column could be added to the “Training Log” indicating the Type of Training performed (e.g., in-person, virtual presentation, e-mail summary with self-review, self-review, other).
These are only a few examples of the types of modifications that could be made and does not represent an complete summary.
If the sponsor has provided a form and asked study sites to use their form, then the sponsor form should be used. If you feel the UU form has an advantage over the sponsor-provided form, you may make a request to the sponsor to use the UU form by submitting the UU form with an explanation of the proposed advantage(s) to the sponsor. Some sponsors may provide template forms which are optional for use and the sponsor may not have a preference for which form is used, as long the required content is captured.
No. Depending on the research, some of these forms may not be applicable, may be unnecessary, and/or may have already been provided for the research.
No. The templates provided in the OQC Toolkit are intended to be used internally by research teams to assist with conducting, tracking and/or monitoring research activities.